|
Stylized depiction of an activated NMDAR. Glutamate is in the glutamate-binding site and glycine is in the glycine-binding site. The, which modulates receptor function when bound to a ligand, is not occupied. NMDARs require the binding of two molecules of glutamate or aspartate and two of glycine. The N-methyl-D-aspartate receptor (also known as the NMDA receptor or NMDAR), is a and found in. The NMDA receptor is one of three types of, the others being the and. 
It is activated when and (or ) bind to it, and when activated it allows to flow through the. The NMDA receptor is very important for controlling and function. The NMDAR is a specific type of. The NMDA receptor is so named because the molecule (NMDA) binds selectively to it, and not to other glutamate receptors. Activation of NMDA receptors results in the opening of an that is nonselective to, with a combined near 0 mV. While the opening and closing of the ion channel is primarily gated by binding, the current flow through the ion channel is voltage dependent. Extracellular magnesium (Mg 2+) and zinc (Zn 2+) ions can bind to specific sites on the receptor, blocking the passage of other cations through the open ion channel. Depolarization of the cell dislodges and repels the Mg 2+ and Zn 2+ ions from the pore, thus allowing a voltage-dependent flow of sodium (Na +) and small amounts of calcium (Ca 2+) ions into the cell and potassium (K +) out of the cell. Ca 2+ flux through NMDARs is thought to be critical in, a cellular mechanism for and. The opening and closing (gating) of the NMDA receptor is complex. While it is primarily a ligand-gated channel, it does display weaker voltage-dependence modulation of the ligand-dependent gating. The ligand gating requires co-activation by two ligands: and either. The voltage-dependence of current through the channel is mainly due to binding of Mg 2+ or Zn 2+ ions to the protein as described above. The activity of the NMDA receptor is affected by many drugs such as (PCP), () and (DXM). The and effects of the drugs and are partially because of their effects on NMDA receptor activity. Contents • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • • In 1972 a possible therapeutic importance of, an derivative, was discovered for treating disorders. Since 1989 memantine has been recognized to be an of the (NMDA receptor), entering the channel of the receptor after it has been activated and thereby blocking the flow of ions. The NMDA receptor is a glutamate and ion channel protein receptor that is activated when glycine and glutamate bind to it. The receptor is a heteromeric complex that. How can the answer be improved? 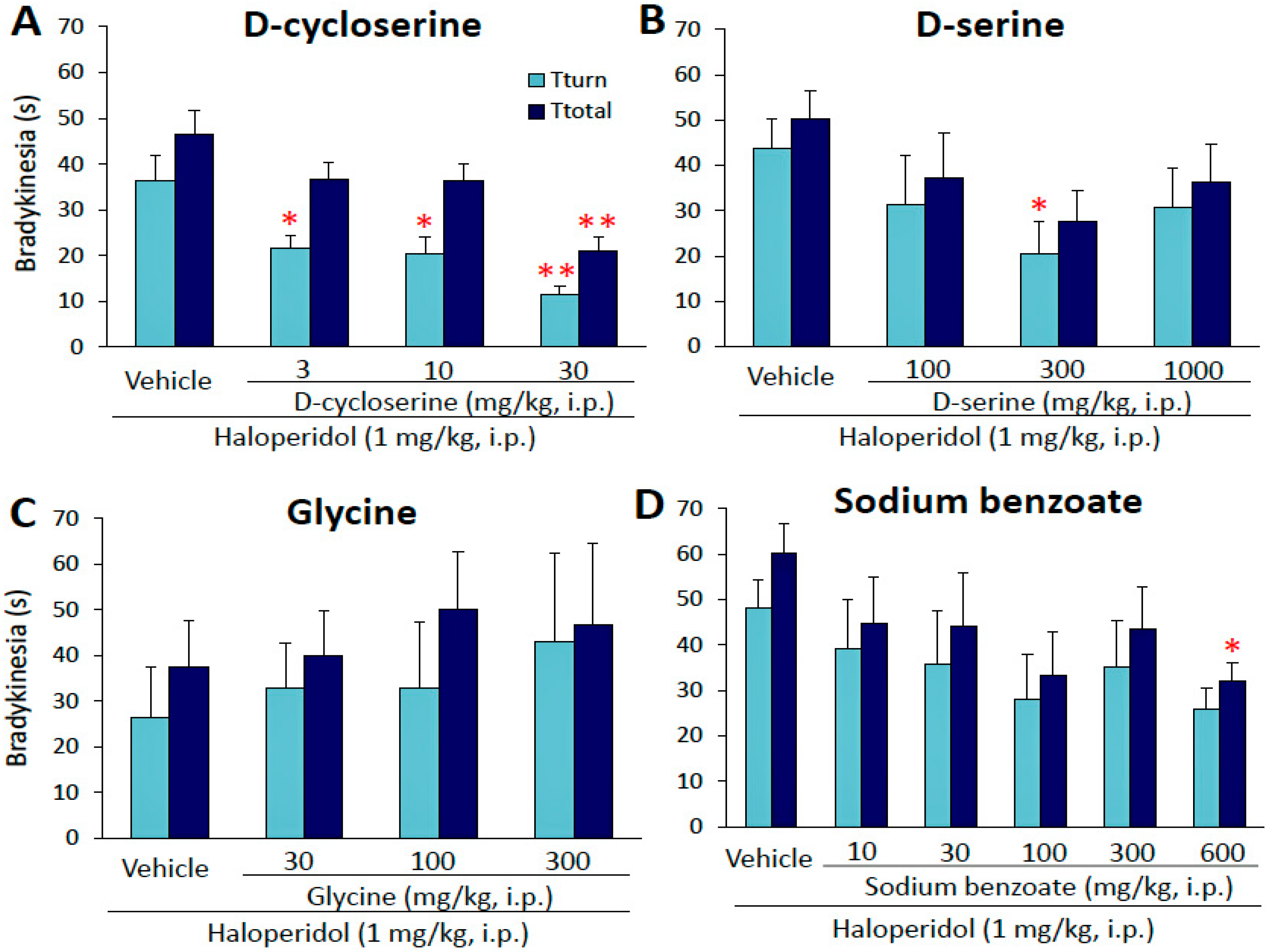
NMDA receptors (NMDARs) are glutamate-gated cation channels with high calcium permeability that play important roles in many aspects of the biology of higher organisms. They are critical for the development of the central nervous system (CNS), generation of rhythms for breathing and locomotion, and. The α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (also known as AMPA receptor. Through the NMDA receptors and the resultant activation of Ca. Article Mechanism of NMDA Receptor Inhibition and Activation Shujia Zhu, 1Richard A. Stein,2 Craig Yoshioka,3 Chia-Hsueh Lee,1,5 April Goehring, Hassane S. The NMDA receptor channels play an important role in and synapse formation underlying memory, learning and formation of neural networks during development in the (CNS). Overactivation of the receptor, causing excessive influx of Ca 2+ can lead to which is implied to be involved in some neurodegenerative disorders. Blocking of NMDA receptors could therefore, in theory, be useful in treating such diseases. The main problem with the development of for is that the physiological NMDA receptor activity is essential for normal neuronal function. To be clinically accepted the antagonists must block excessive activation without blocking the normal function. History [ ] The discovery of NMDA receptors was followed by the synthesis and study of N-Methyl-D-aspartic acid (NMDA) in the 1960s by Jeff Watkins and colleagues. In the early 1980s, NMDA receptors were shown to be involved in several central synaptic pathways. Receptor subunit selectivity was discovered in the early 1990s, which led to recognition of a new class of compounds that selectively inhibit the subunit. These findings led to vigorous campaign in the pharmaceutical industry. From this it was considered that NMDA receptors were associated with a variety of such as,,, and other CNS disorders. A fortuitous finding was made in 1968 when a woman was taking as flu medicine and experienced remarkable remission of her Parkinson's symptoms. This finding, reported by Scawab et al., was the beginning of of adamantane derivatives in the context of diseases affecting the CNS. Before this finding, memantine, another adamantane derivative, had been synthesized by Eli Lilly and Company in 1963. The purpose was to develop drug, but it showed no such. It was not until 1972 that a possible therapeutic importance of memantine for treating neurodegenerative disorders was discovered. From 1989 memantine has been recognized to be an uncompetitive antagonist of the NMDA receptor. NMDA receptor [ ]. Figure 2: Transmembrane region of NR1 (left) and NR2B (right) subunits of NMDA receptor. The NMDA receptor is a and protein receptor that is activated when and glutamate bind to it. The receptor is a heteromeric complex that interacts with multiple intracellular proteins by three different subunits: NR1, NR2 and NR3. NR1 has eight different subunits generated by alternative splicing from a single gene. There are four different NR2 subunits (A-D) and late in the 20 century NR3A and NR3B subunits have been reported. Six separate genes encode for NR2 and NR3. All the subunits share a common membrane topology that is dominated by a large extracellular N-terminus, a membrane region comprising three transmembrane segments, a re-entrant pore loop, an extracellular loop between the transmembrane segments that are structurally not well known, and an intracellular C-terminus, which are different in size depending on the subunit and provide multiple sites of interaction with many intracellular proteins. Figure 1 shows a basic structure of NR1/NR2 subunits that forms the for memantine, Mg 2+,, and amantadine. Mg 2+ block the NMDA receptor channels in voltage dependent manner but they are highly permeable to Ca 2+. Activation of the receptor depends on glutamate binding, or glycine binding at its NR1-linked binding site and -mediated of the postsynaptic membrane, which relieves the voltage-dependent channel block by Mg 2+. Activation and opening of the receptors channel thus allows the flow of K +, Na + and Ca 2+ ions, and the influx of Ca 2+ triggers intracellular signaling pathways. Allosteric receptor binding sites for zinc, proteins and the polyamines spermidine and spermine are also modulators for the NMDA receptor channels. The NR2B subunit has been involved in modulating activity such as learning, memory, processing and feeding behaviors, as well as being implicated in number of human derangement. The basic structure and functions associated with the NMDA receptor can be attributed to the NR2B subunit. For example, the glutamate binding site and the control of the Mg 2+ block are formed by the NR2B subunit. The high affinity sites for glycine are also exclusively displayed by the NR1/NR2B receptor. NR1/NR2B transmembrane segments are considered to be the part of the receptor that forms the binding pockets for uncompetitive NMDA receptor antagonists, but the transmembrane segments structures are not fully known as stated above. It is claimed that three binding sites within the receptor, A644 on the NR2B subunit and A645 and N616 on the NR1 subunit, are important for binding of memantine and related compounds as seen in figure 2. The NMDA receptor forms a between two GluN1 and two GluN2 subunits (the subunits were previously denoted as NR1 and NR2), two obligatory NR1 subunits and two regionally localized NR2 subunits. A related family of NR3 A and B subunits have an inhibitory effect on receptor activity. Multiple receptor with distinct brain distributions and functional properties arise by selective splicing of the NR1 transcripts and differential expression of the NR2 subunits. Each receptor subunit has modular design and each structural module also represents a functional unit: • The contains two globular structures: a modulatory domain and a -binding domain. NR1 subunits bind the co-agonist glycine and NR2 subunits bind the neurotransmitter glutamate. • The agonist-binding module links to a membrane domain, which consists of three transmembrane segments and a re-entrant loop reminiscent of the selectivity filter of. • The membrane domain contributes residues to the channel pore and is responsible for the receptor's high-unitary, high-calcium permeability, and voltage-dependent magnesium block. • Each subunit has an extensive cytoplasmic domain, which contain residues that can be directly modified by a series of and, as well as residues that interact with a large number of structural, adaptor, and scaffolding proteins. The glycine-binding modules of the NR1 and NR3 subunits and the glutamate-binding module of the NR2A subunit have been expressed as soluble proteins, and their three-dimensional structure has been solved at atomic resolution. This has revealed a common fold with amino acid-binding bacterial proteins and with the glutamate-binding module of AMPA-receptors and kainate-receptors. Mechanism of action [ ] Overactivation of NMDA receptors, relieving the Mg 2+ block and causing excessive influx of Ca 2+ can lead to excitotoxicity. Excitotoxicity is implied to be involved in some neurodegenerative disorders such as Alzheimer's disease, Parkinson's disease and Huntington's disease. Blocking of NMDA receptors could therefore, in theory, be useful in treating such diseases. It is, however, important to preserve physiological NMDA receptor activity while trying to block its excessive, excitotoxic activity. This can possibly be achieved by uncompetitive antagonists, blocking the receptors ion channel when excessively open. Uncompetitive NMDA receptor antagonists, or channel blockers, enter the channel of the NMDA receptor after it has been activated and thereby block the flow of ions. MK-801, ketamine, amantadine and memantine are examples of such antagonists, see figure 1. The off-rate of an antagonist from the receptors channel is an important factor as too slow off-rate can interfere with normal function of the receptor and too fast off-rate may give ineffective blockade of an excessively open receptor. Memantine is an example of an uncompetitive channel blocker of the NMDA receptor, with a relatively rapid off-rate and low affinity. At physiological pH its amine group is positively charged and its block of the channel is voltage-dependent. It thereby mimics the physiological function of Mg 2+ as channel blocker. Memantine only blocks NMDA receptor associated channels during prolonged activation of the receptor, as it occurs under excitotoxic conditions, by replacing magnesium at the binding site. During normal receptor activity the channels only stay open for several milliseconds and under those circumstances memantine is unable to bind within the channels and therefore doesn't interfere with normal synaptic activity. Variants [ ] GluN1 [ ] There are eight variants of the subunit produced by alternative splicing of: • NR1-1a, NR1-1b; NR1-1a is the most abundantly expressed form. • NR1-2a, NR1-2b; • NR1-3a, NR1-3b; • NR1-4a, NR1-4b; GluN2 [ ]. NR2 subunit in vertebrates (left) and invertebrates (right). Ryan et al., 2008 While a single NR2 subunit is found in invertebrate organisms, four distinct isoforms of the NR2 subunit are expressed in vertebrates and are referred to with the nomenclature NR2A through NR2D (encoded by,,, ). Strong evidence shows that the genes encoding the NR2 subunits in vertebrates have undergone at least two rounds of gene duplication. They contain the binding-site for the. More importantly, each NR2 subunit has a different intracellular C-terminal domain that can interact with different sets of signalling molecules. Unlike NR1 subunits, NR2 subunits are expressed differentially across various cell types and control the electrophysiological properties of the NMDA receptor. One particular subunit, NR2B, is mainly present in immature neurons and in extrasynaptic locations, and contains the binding-site for the selective inhibitor. NR2B to NR2A switch [ ]. The time course of NR2B-NR2A switch in human cerebellum. Bar-Shira et al., 2015 Whereas is predominant in the early postnatal brain, the number of NR2A subunits grows, and eventually subunits outnumber NR2B. This is called the NR2B-NR2A developmental switch, and is notable because of the different kinetics each NR2 subunit lends to the receptor. For instance, greater ratios of the NR2B subunit leads to NMDA receptors which remain open longer compared to those with more NR2A. This may in part account for greater memory abilities in the immediate postnatal period compared to late in life, which is the principle behind genetically altered '. The detailed time course of this switch in the human cerebellum has been estimated using expression microarray and RNA seq and is shown in the figure on the right. There are three hypothetical models to describe this switch mechanism: • Increase in synaptic NR2A along with decrease in NR2B • Extrasynaptic displacement of NR2B away from the synapse with increase in NR2A • Increase of NR2A diluting the number of NR2B without the decrease of the latter. The NR2B and NR2A subunits also have differential roles in mediating neuronal death. The developmental switch in subunit composition is thought to explain the developmental changes in NMDA neurotoxicity. Disruption of the gene for NR2B in mice causes perinatal, whereas the disruption of NR2A gene produces viable mice, although with impaired hippocampal plasticity. One study suggests that may play a role in the NMDA receptor maturation by increasing the subunit mobility. NR2B to NR2C switch [ ] Granule cell precursors (GCPs) of the cerebellum, after undergoing symmetric cell division in the external granule-cell layer (EGL), migrate into the internal granule-cell layer (IGL) where they downregulate NR2B and activate NR2C, a process that is independent of neuregulin beta signaling through ErbB2 and ErbB4 receptors. Role in excitotoxicity [ ] NMDA receptors have been implicated by a number of studies to be strongly involved with. Because NMDA receptors play an important role in the health and function of, there has been much discussion on how these receptors can affect both cell survival and cell death. Recent evidence supports the hypothesis that overstimulation of extrasynaptic NMDA receptors has more to do with excitotoxicity than stimulation of their counterparts. In addition, while stimulation of extrasynaptic NMDA receptors appear to contribute to cell death, there is evidence to suggest that stimulation of synaptic NMDA receptors contributes to the health and longevity of the cell. There is ample evidence to support the dual nature of NMDA receptors based on location, and the hypothesis explaining the two differing mechanisms is known as the 'localization hypothesis'. Differing cascade pathways [ ] In order to support the localization hypothesis, it would be necessary to show differing are activated by NMDA receptors based on its location within the cell membrane. Experiments have been designed to stimulate either synaptic or non-synaptic NMDA receptors exclusively. These types of experiments have shown that different pathways are being activated or regulated depending on the location of the signal origin. Many of these pathways use the same, but are regulated oppositely by NMDARs depending on its location. For example, synaptic NMDA excitation caused a decrease in the intracellular concentration of p38 mitogen-activated protein kinase (). Extrasynaptic stimulation NMDARs regulated p38MAPK in the opposite fashion, causing an increase in intracellular concentration. Experiments of this type have since been repeated with the results indicating these differences stretch across many pathways linked to cell survival and excitotoxicity. Two specific proteins have been identified as a major pathway responsible for these different cellular responses, and Jacob. ERK1/2 is responsible for phosphorylation of Jacob when excited by synaptic NMDARs. This information is then. Phosphorylation of Jacob does not take place with extrasynaptic NMDA stimulation. This allows the in the nucleus to respond differently based in the phosphorylation state of Jacob. Neural plasticity [ ] NMDA receptors are also associated with synaptic plasticity. The idea that both synaptic and extrasynaptic NMDA receptors can affect (LTP) and (LTD) differently has also been explored. Experimental data suggest that extrasynaptic NMDA receptors inhibit LTP while producing LTD. Inhibition of LTP can be prevented with the introduction of a. A that usually induces LTP with synaptic NMDARs, when applied selectively to extrasynaptic NMDARs produces a LTD. Experimentation also indicates that extrasynaptic activity is not required for the formation of LTP. In addition, both synaptic and extrasynaptic are involved in expressing a full LTD. Role of differing subunits [ ] Another factor that seems to affect NMDAR induced toxicity is the observed variation in makeup. NMDA receptors are heterotetramers with two GluN1 subunits and two variable subunits. Two of these variable subunits, GluN2A and GluN2B, have been shown to preferentially lead to cell survival and cell death cascades respectively. Although both subunits are found in synaptic and extrasynaptic NMDARs there is some evidence to suggest that the GluN2B subunit occurs more frequently in extrasynaptic receptors. This observation could help explain the dualistic role that NMDA receptors play in excitotoxicity. Despite the compelling evidence and the relative simplicity of these two theories working in tandem, there is still disagreement about the significance of these claims. Some problems in proving these theories arise with the difficulty of using pharmacological means to determine the subtypes of specific NMDARs. In addition, the theory of subunit variation does not explain how this effect might predominate, as it is widely held that the most common tetramer, made from two GluN1 subunits and one of each subunit GluN2A and GluN2B, makes up a high percentage of the NMDARs. Excitotoxicity in a clinical setting [ ] Excitotoxicity has been thought to play a role in the degenerative properties of conditions since the late 1950s. NMDA receptors seem to play an important role in many of these degenerative diseases affecting the brain. Most notably excitotoxic events involving NMDA receptors have been linked to Alzheimer's disease and Huntington's disease as well as with other medical conditions such as strokes and epilepsy. Treating these conditions with one of the many known NMDA receptor antagonists, however, lead to a variety of unwanted side effects, some of which can be quite severe. These side effects are, in part, observed because the NMDA receptors do not just signal for cell death but also play an important role in its vitality. Treatment for these conditions might be found in blocking NMDA receptors not found at the synapse. Ligands [ ] Agonists [ ]. The major endogenous agonist of the glycine co-agonist site of the NMDAR. Activation of NMDA receptors requires binding of or (aspartate does not stimulate the receptors as strongly). In addition, NMDARs also require the binding of the co- for the efficient opening of the ion channel, which is a part of this receptor. Has also been found to co-agonize the NMDA receptor with even greater potency than glycine. It is produced by, and is enriched in the same areas as NMDA receptors. Removal of D-serine can block NMDA-mediated excitatory neurotransmission in many areas. Recently, it has been shown that D-serine can be released both by neurons and astrocytes to regulate NMDA receptors. NMDA receptor (NMDAR)-mediated currents are directly related to membrane depolarization. NMDA agonists therefore exhibit fast unbinding kinetics, increasing channel open probability with depolarization. This property is fundamental to the role of the NMDA receptor in and, and it has been suggested that this channel is a biochemical substrate of, where it can act as a coincidence detector for membrane depolarization and synaptic transmission. Figure 6: Chemical structure of neramexane, second generation memantine derivative. An example of memantine derivative is which was discovered by studying number of aminoalkyl, with memantine as the template, as NMDA receptor antagonists. Neramexane, which can be seen in figure 6, binds to the same site as memantine within the NMDA receptor associated channel and with comparable affinity. It does also show very similar bioavailability and blocking kinetics as memantine. Neramexane went to for four indications, including Alzheimer's disease. Partial agonists [ ]. (NMDA), a synthetic partial agonist of the main site of the NMDAR. (NMDA), which the NMDA receptor was named after, is a partial agonist of the active or glutamate recognition site. 3,5-Dibromo-L-phenylalanine, a naturally occurring halogenated derivative of, is a weak partial NMDA receptor agonist acting on the glycine site. 3,5-Dibromo-L-phenylalanine has been proposed a novel therapeutic drug candidate for treatment of neuropsychiatric disorders and diseases such as, and neurological disorders such as and. Other weak partial agonists of the glycine site of the NMDA receptor such as (GLYX-13) and (NRX-1074) are now viewed for the development of new drugs with antidepressant and analgesic effects without obvious psychotomimetic activities. Examples [ ] • (ACC) – synthetic glycine site partial agonist • () – naturally occurring glycine site partial agonist found in • – synthetic glycine site weak partial agonist • – synthetic glutamate site partial agonist • (NMDA) – synthetic glutamate site partial agonist Positive allosteric modulators include: • (NRX-1074) – synthetic weak partial agonist of an allosteric site of the glycine site • (GLYX-13) – synthetic weak partial agonist of an allosteric site of the glycine site Antagonists [ ]. A synthetic general anesthetic and one of the best-known NMDAR antagonists. Antagonists of the NMDA receptor are used as for animals and sometimes humans, and are often used as due to their properties, in addition to their unique effects at elevated dosages such as. When certain NMDA receptor antagonists are given to rodents in large doses, they can cause a form of called. NMDA receptor antagonists that have been shown to induce Olney's lesions include,, and (a metabolite of ), as well as some NMDA receptor antagonists used only in research environments. So far, the published research on Olney's lesions is inconclusive in its occurrence upon human or monkey brain tissues with respect to an increase in the presence of NMDA receptor antagonists. Most NMDAR antagonists are or of the channel pore or are antagonists of the glycine co-regulatory site rather than antagonists of the active/glutamate site. Figure 7: Nitroglycerin donate ONO 2 group that leads to second generation memantine analog, nitromemantine. Include: • – endogenous weak negative allosteric modulator • – naturally occurring negative allosteric modulators of the polyamine site found in Modulators [ ] Examples [ ] The NMDA receptor is modulated by a number of and compounds: • have been shown to have a similar effect to polyamines, and this may explain their neurotoxic effect. • regulates the amount of -containing NMDA receptors on the synaptic membrane, thus affecting. • do not directly activate NMDA receptors, but instead act to potentiate or inhibit glutamate-mediated responses. • modulates NMDA function through and. Significantly enhancing in the. • kinase enhances NMDA receptor currents. •, and not only pass through the NMDA receptor channel but also modulate the activity of NMDA receptors. • and generally block NMDA current activity in a noncompetitive and a voltage-independent manner. However zinc may potentiate or inhibit the current depending on the neural activity. • 2+ is a potent NMDAR antagonist. Presynaptic deficits resulting from Pb 2+ exposure during synaptogenesis are mediated by disruption of NMDAR-dependent BDNF signaling. • Proteins of the class I are endogenous negative regulators of NMDAR-mediated currents in the adult hippocampus, and are required for appropriate NMDAR-induced changes in trafficking and NMDAR-dependent and and. • The activity of NMDA receptors is also strikingly sensitive to the changes in, and partially inhibited by the ambient concentration of H + under physiological conditions. The level of inhibition by H + is greatly reduced in receptors containing the NR1a subtype, which contains the positively charged insert Exon 5. The effect of this insert may be mimicked by positively charged polyamines and aminoglycosides, explaining their mode of action. • NMDA receptor function is also strongly regulated by chemical reduction and oxidation, via the so-called 'redox modulatory site.' Through this site, reductants dramatically enhance NMDA channel activity, whereas oxidants either reverse the effects of reductants or depress native responses. It is generally believed that NMDA receptors are modulated by endogenous redox agents such as,, and the essential nutrient. Development of NMDA receptor antagonist [ ] The main problem with the development of NMDA antagonist for neuroprotection is that the physiological NMDA receptor activity is essential for normal neuronal function. Complete blocking of all NMDA receptor activity therefore results in adverse side effects such as, agitation and. To be clinically accepted the NMDA receptor antagonist must block excessive activation without blocking the normal function. Figure 3 shows simplified models of various types of NMDA receptor antagonists, which will be discussed further. Competitive NMDA receptor antagonists [ ] NMDA receptor antagonists, which were developed first, are not a good option because they compete and bind to the same site (NR2 subunit) on the receptor as the agonist, glutamate, and therefore block normal function also. They will block healthy areas of the brain prior to having an impact on pathological areas, because healthy areas contain lower levels of than pathological areas. These antagonists can be displaced from the receptor by high concentration of glutamate which can exist under excitotoxic circumstances. Uncompetitive NMDA receptor antagonists [ ]. Figure 4: The chemical structures of MK-801, phencyclidine and ketamine, high affinity uncompetitive NMDA receptor antagonists. Uncompetitive NMDA receptor antagonists however block within the ion channel at the Mg 2+ site (pore region) and in that way prevent excessive influx of Ca 2+. Uncompetitive block refers to a type of block that increased concentration of glutamate cannot overcome and is dependent upon prior activation of the receptor by the agonist, i.e. It only enters the channel when it is opened by agonist. The Mg 2+ block itself is too transient and flickery and therefore doesn't block excessive Ca 2+ influx to the extent necessary to prevent neurological toxicity. High affinity antagonists for the Mg 2+ site are on the other hand good excitotoxicity blockers, such as MK-801. They block open ion channels but the problem is when the ion channels close they get trapped inside resulting in undesirable side effects because of blocking normal as well as excessive activity. MK-801 cannot be given to humans because of that long dwell time in the channels and its blocking causes drowsiness and even coma. It's therefore considered to be clinically unacceptable., which has slightly shorter dwell time but still too excessive, causes hallucination and is therefore not a good agent either for neurodegenerative diseases. Ketamine is another example of drug with slightly shorter dwell time but still excessive and it is used as. Chemical structures of MK-801, Phencyclidine and Ketamine can be seen in figure 4. Memantine and related compounds [ ]. Figure 5: Chemical structures of memantine (right) and amantadine (left). Because of these adverse side effects of high affinity blockers the search for clinically successful NMDA receptor antagonists for neurodegenerative diseases continued and focused on developing low affinity blockers. However the affinity could not be too low and dwell time not too short (as seen with Mg 2+) where membrane depolarization relieves the block. The discovery was thereby development of uncompetitive antagonist with longer dwell time than Mg 2+ in the channel but shorter than MK-801. That way the drug obtained would only block excessively open NMDA receptor associated channels but not normal neurotransmission. Memantine is that drug. It is a derivative of amantadine which was first an anti-influenza agent but was later discovered by coincidence to have efficacy in Parkinson's disease. Chemical structures of memantine and amantadine can be seen in figure 5. The compound was first thought to be or but was later found to be an NMDA receptor antagonist. Memantine is the first drug approved for treatment of severe and more advanced, which for example anticholinergic drugs do not do much good for. It helps recovery of synaptic function and in that way improves impaired memory and learning. In 2015 memantine is also in trials for therapeutic importance in additional neurological disorders. Many second-generation memantine derivatives have been in development that may show even better neuroprotective effects, where the main thought is to use other safe but effective modulatory sites on the NMDA receptor in addition to its associated ion channel. Structure activity relationship (SAR) [ ]. Figure 8: Structure activity relationship (SAR) of amantadine and related compounds Memantine (1-amino-3,5-dimethyladamantane) is an aminoalkyl cyclohexane derivative and an atypical drug compound with non-planar, three dimensional tricyclic structure. Figure 8 shows SAR for aminoalkyl cyclohexane derivative. Memantine has several important features in its structure for its effectiveness: • Three-ring structure with a bridgehead amine, -NH 2 • The -NH 2 group is protonated under physiological pH of the body to carry a positive charge, -NH 3+ • Two methyl (CH 3) side groups which serve to prolong the dwell time and increase stability as well as affinity for the NMDA receptor channel compared with amantadine (1-adamantanamine). Despite the small structural difference between memantine and amantadine, two adamantane derivatives, the affinity for the binding site of NR1/NR2B subunit is much greater for memantine. In measurements memantine has an of (2.3+0.3) µM while amantadine has an IC 50 of (71.0+11.1) µM. The binding site with the highest affinity is called the dominant binding site. It involves a connection between the amine group of memantine and the NR1-N161 binding pocket of the NR1/NR2B subunit. The methyl side groups play an important role in increasing the affinity to the open NMDA receptor channels and making it a much better neuroprotective drug than amantadine. The binding pockets for the methyl groups are considered to be at the NR1-A645 and NR2B-A644 of the NR1/NR2B. The binding pockets are shown in figure 2. Memantine binds at or near to the Mg 2+ site inside the NMDA receptor associated channel. The -NH 2 group on memantine, which is protonated under physiological pH of the body, represents the region that binds at or near to the Mg 2+ site. Adding two methyl groups to the -N on the memantine structure has shown to decrease affinity, giving an IC 50 value of (28.4+1.4) µM. Second generation derivative of memantine; Nitromemantine [ ] Several derivatives of Nitromemantine, a second-generation derivative of memantine, have been synthesized in order to perform a detailed (SAR) of these novel drugs. One class, containing a nitro (NO 2) group opposite to the bridgehead amine (NH 2), showed a promising outcome. Nitromemantine utilizes memantine binding site on the NMDA receptor to target the NO x (X= 1 or 2) group for interaction with the S- nitrosylation/redox site external to the memantine binding site. Lengthening the side chains of memantine compensates for the worse drug affinity in the channel associated with the addition of the –ONO 2 group Therapeutic application [ ] Excitotoxicity is implied to be involved in some neurodegenerative disorders such as Alzheimer's disease, Parkinson's disease, Huntington's disease and. Blocking of NMDA receptors could therefore, in theory, be useful in treating such diseases. It is, however, important to preserve physiological NMDA receptor activity while trying to block its excessive, excitotoxic activity. This can possibly be achieved by uncompetitive antagonists, blocking the receptors ion channel when excessively open Memantine is an example of uncompetitive NMDA receptor antagonist that has approved indication for the neurodegenerative disease Alzheimer's disease. In 2015 memantine is still in clinical trials for additional neurological diseases. Receptor modulation [ ] The NMDA receptor is a non-specific cation channel that can allow the passage of Ca 2+ and Na + into the cell and K + out of the cell. The (EPSP) produced by activation of an NMDA receptor increases the concentration of Ca 2+ in the cell. The Ca 2+ can in turn function as a in various. However, the NMDA receptor cation channel is blocked by Mg 2+ at resting membrane potential. Magnesium unblock is not instantaneous, to unblock all available channels, the postsynaptic cell must be depolarized for a sufficiently long period of time (in the scale of milliseconds). Therefore, the NMDA receptor functions as a 'molecular '. Its ion channel opens only when the following two conditions are met: glutamate is bound to the receptor, and the postsynaptic cell is depolarized (which removes the Mg 2+ blocking the channel). This property of the NMDA receptor explains many aspects of (LTP) and. NMDA receptors are modulated by a number of endogenous and exogenous compounds and play a key role in a wide range of (e.g., ) and processes (e.g., ). Clinical significance [ ] NMDAR antagonists like,,,,, and are used as. These and similar drugs like and also produce,, and effects and are used as. NMDAR inhibitors, including ketamine, (JNJ-54135419), (GLYX-13), (NRX-1074), (AV-101), and (CERC-301, MK-0657), are under development for the treatment of, including and. In addition, ketamine is already employed for this purpose as an off-label therapy in some clinics., a low-trapping NMDAR antagonist, is approved in the and for the treatment of moderate-to-severe Alzheimer's disease, and has now received a limited recommendation by the UK's for patients who fail other treatment options. Cochlear NMDARs are the target of intense research to find pharmacological solutions to treat. NMDARs are associated with a rare disease,, that usually occurs due to cross-reactivity of antibodies produced by the immune system against ectopic brain tissues, such as those found in. These are known as. Compared to like, the NMDAR antagonist phencyclidine can produce a wider range of symptoms that resemble schizophrenia in healthy volunteers, in what has led to the. Experiments in which rodents are treated with NMDA receptor antagonist are today the most common model when it comes to testing of novel schizophrenia therapies or exploring the exact mechanism of drugs already approved for treatment of schizophrenia. NMDAR antagonists, for instance,,, and have been extensively investigated for the treatment of -mediated in situations like and, but were unsuccessful in. See also [ ].
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
May 2018
Categories |
 RSS Feed
RSS Feed
